Which out of ammonia (NH3) and NF3 has higher dipole moment and why? - Sarthaks eConnect | Largest Online Education Community

Sketch NH3 to show the correct molecular geometry and bond dipoles. Then state whether it is polar or nonpolar. | Homework.Study.com

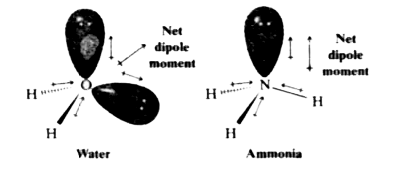
Is NH3 polar Or Nonpolar? - nh3 Intermolecular Forces, nh3 charge, nh3 bond angle, detailed facts? - chemwhite.com

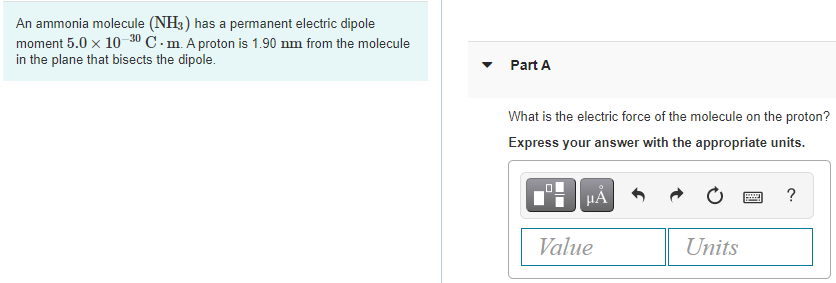
What is the correct dipole moment of $N{{H}_{3}}$ and $N{{F}_{3}}$ respectively?(A)- $4.90\\times {{10}^{-30}}$ C m and $0.80\\times {{10}^{-30}}$ C m(B)- $0.80\\times {{10}^{-30}}$ C m and $4.90\\times {{10}^{-30}}$ C m(C)- $4.90\\times {{10}^{-30}}$ C

Dipole Moment as a Possible Diagnostic Descriptor of the Conformational Isomerism of the Ammonia Molecule | Semantic Scholar

Dipole distribution in the two polymorphs of ammoniaborane viewed along... | Download Scientific Diagram
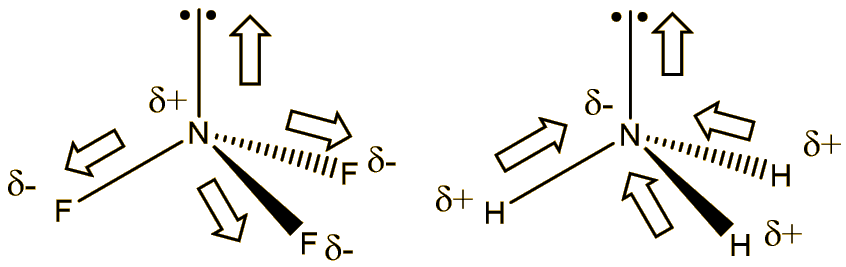
Why is the moment dipole for ammonia (1.64D substantially higher than that of ammonia trifluoride (0.24D) although the latter contains the highly electronegative flourine? - Quora
Why does ammonia has induced dipole induced dipole attraction? Whats the difference between induced dipole induced dipole, dipole dipole and bonds? - Quora

Can you explain why NH3 has such a large dipole moment compared with NF3? Show work. | Homework.Study.com

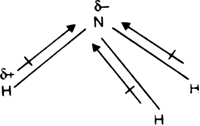
Although flourine is more electronegative than nitrogen, the resultant dipole moment of ammonia is greater than nitrogen trifluoride Explain - Chemistry - Chemical Bonding and Molecular Structure - 6229927 | Meritnation.com










![Which out of \\[N{H_3}\\] and $N{F_3}$ has a higher dipole moment and why? Which out of \\[N{H_3}\\] and $N{F_3}$ has a higher dipole moment and why?](https://www.vedantu.com/question-sets/a32afb56-fc67-43ca-8a5c-e06f6fe2b15f5994708008049533229.png)
