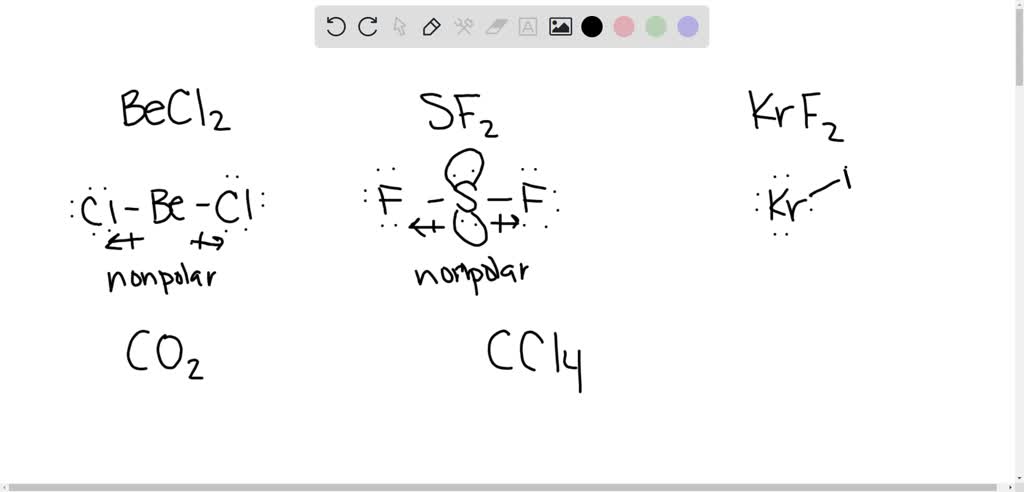
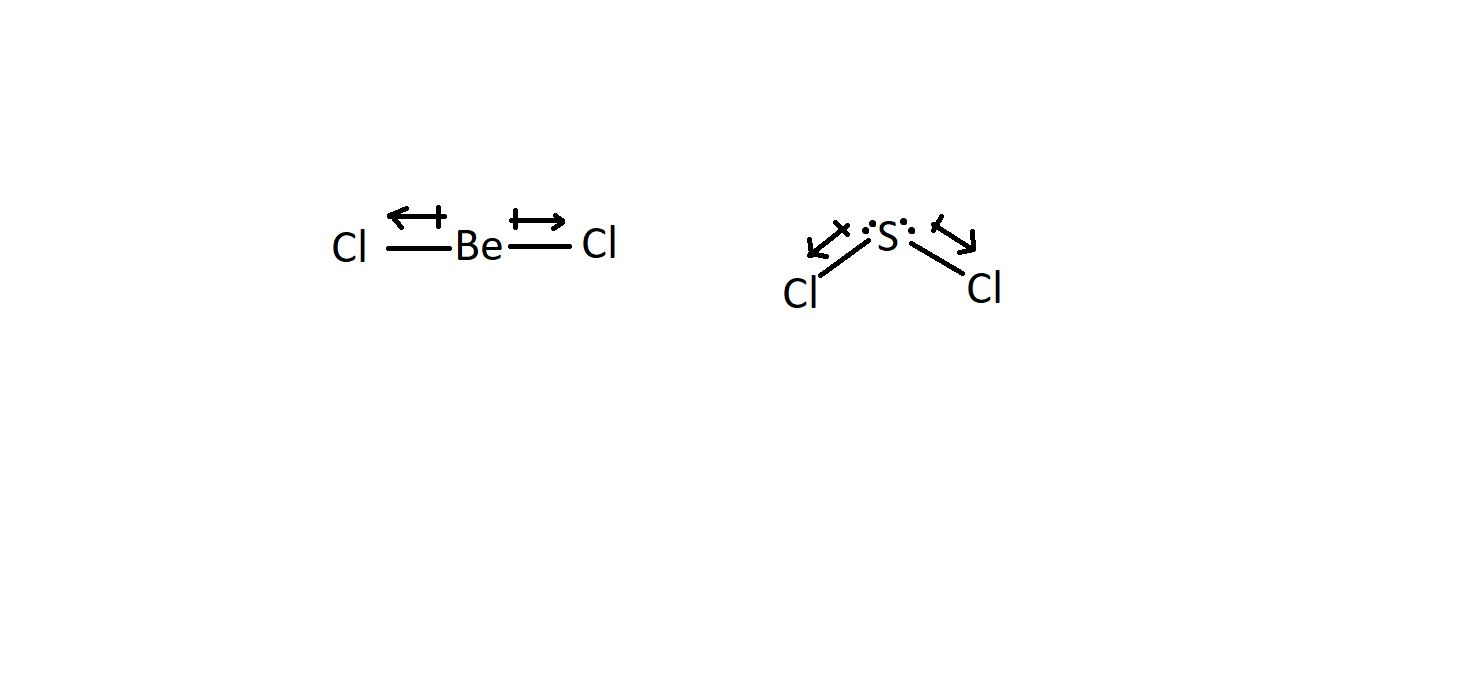
SOLVED: Which of the following molecules has a net dipole moment (which one is polar)? A) BeCl2 B) SF2 C) KrF2 D) CO2 E) CCl4

Identify the option where all four molecules posses permanent dipole moment room temperature (A) BF3, O3, SF6, XeF6 (B) BeCl2, CO2, BC13, CHC1z (B) SO2, C6HzC1, H2Se, BrF5 (B) NO2, NH3, POCI3,
Welcome to Chem Zipper.com......: What is hydrolysis product of BeCl2 with excess water at room temperature?
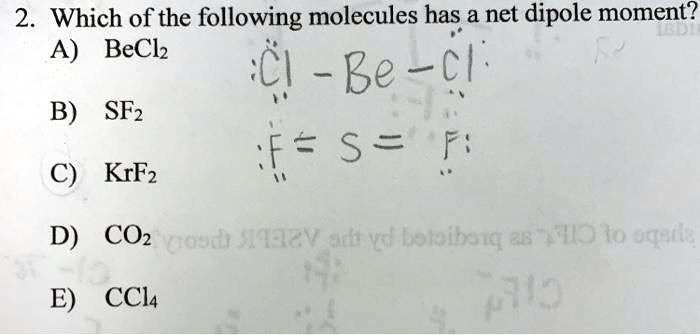
SOLVED: Which of the following molecules has a net dipole moment? A) BeCl2 (Be-Cl) B) SF2 (F-S-F) C) KrF2 D) CO2 E) CCl4

Must polar bonds give rise to polar molecules? And, why is water a polar molecule? | anhourofchemaday

Question 17 Give reason the following: @ Dipole moment of BF3 is zero but ammonia has a dipole moment. (6) Cuci is covalent than Naci. C) LiCl is covalent than NaCl. (

Dipole moment of so2, h2o, ccl4, chcl3, cis & trans alkenes, co2, nh3, bf3, ch4 & organic compounds - YouTube
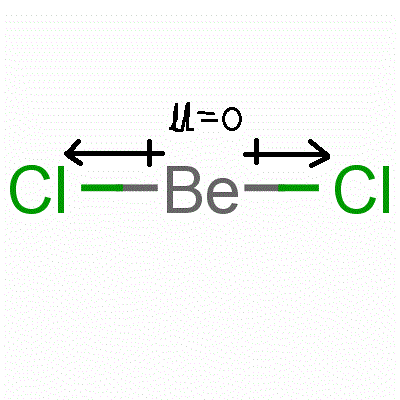










![Telugu] Assertion (A) : BeCl2, molecule is linear in shape.Reason (R) Telugu] Assertion (A) : BeCl2, molecule is linear in shape.Reason (R)](https://d10lpgp6xz60nq.cloudfront.net/ss/web-overlay-thumb/10812639.webp)

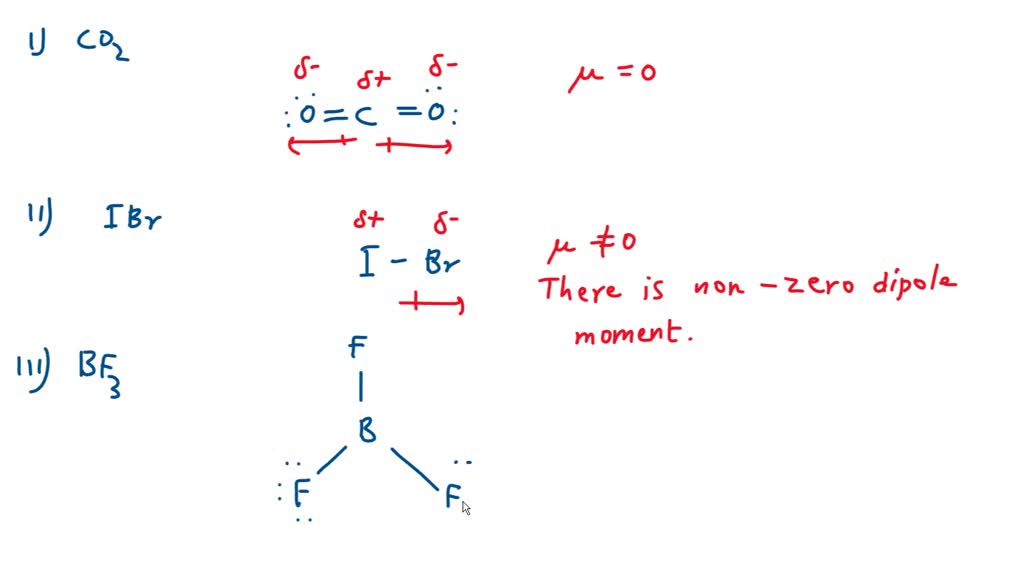


![Best Overview: Is BeCl2 Polar or Nonpolar [No#1] - Science Education and Tutorials Best Overview: Is BeCl2 Polar or Nonpolar [No#1] - Science Education and Tutorials](https://sciedutut.com/wp-content/uploads/2021/05/Is-BeCl2-Polar-or-Non-Polar-2-1024x493.png)