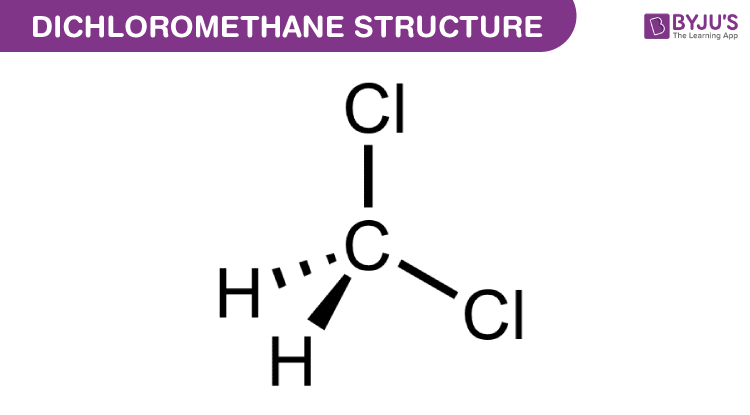
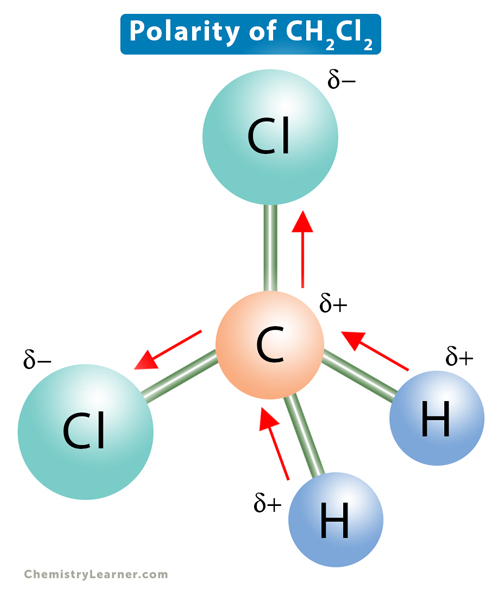
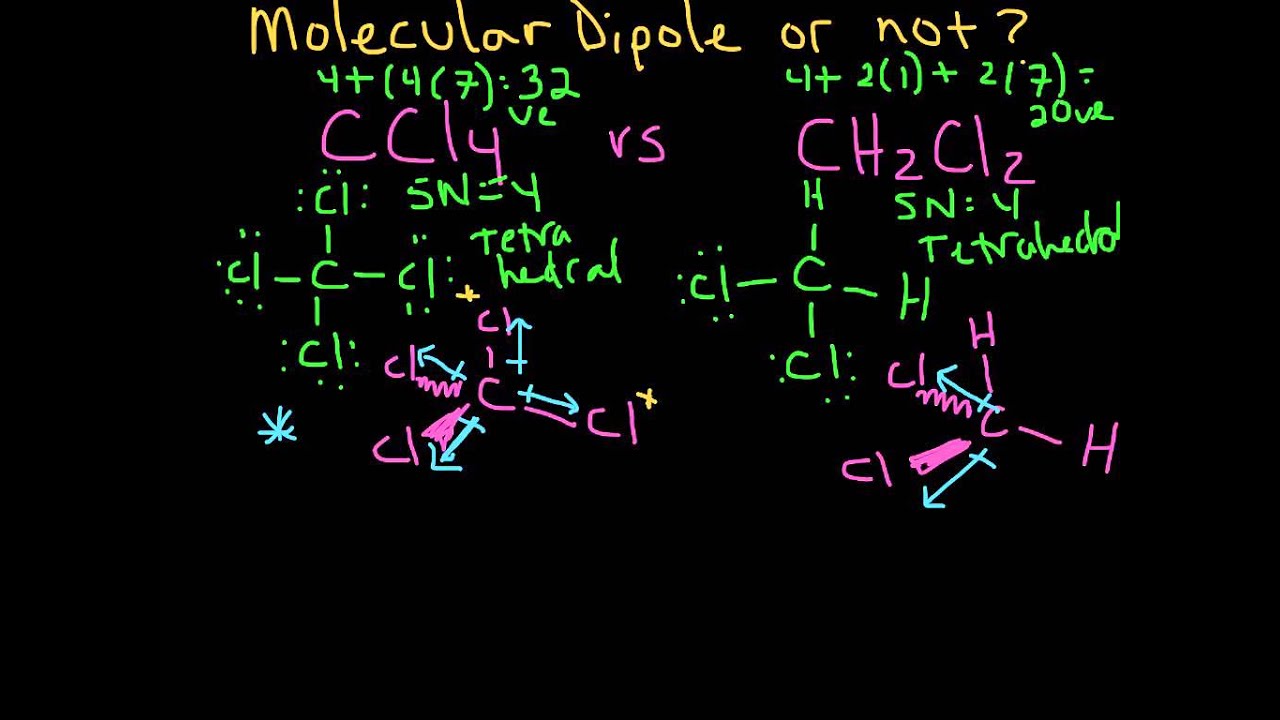halides - Why does chloromethane have a larger dipole moment than chloroform? - Chemistry Stack Exchange

Arrange the following in the increasing order of property indicated: dichromomethane, chloroform, carbon tetrachloride ( Increasing order of dipole moment) - Chemistry - Haloalkanes and Haloarenes - 11375363 | Meritnation.com