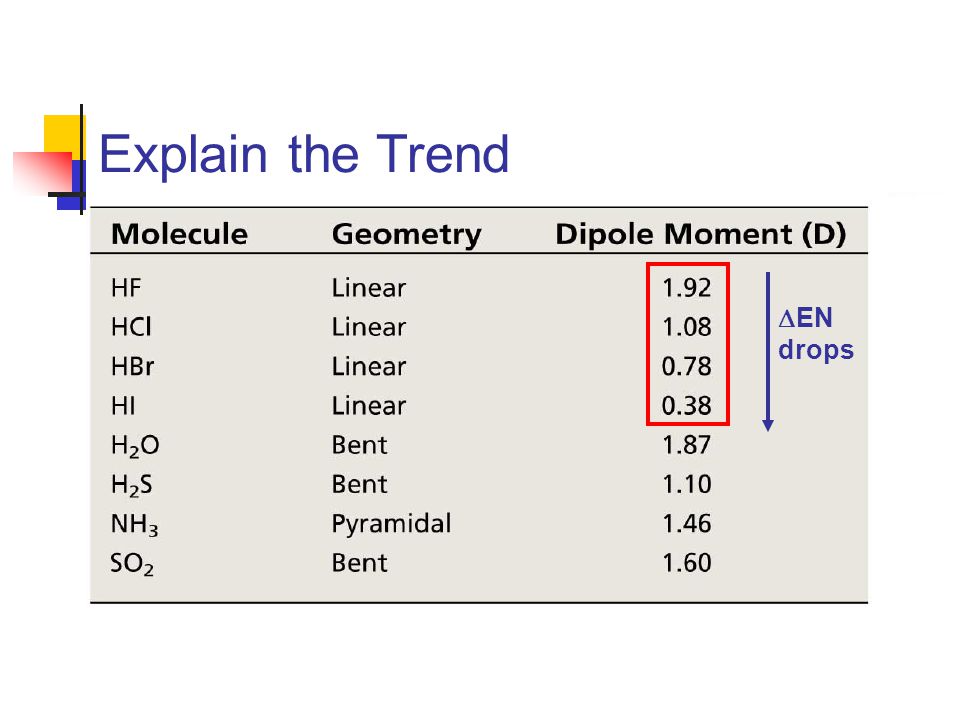
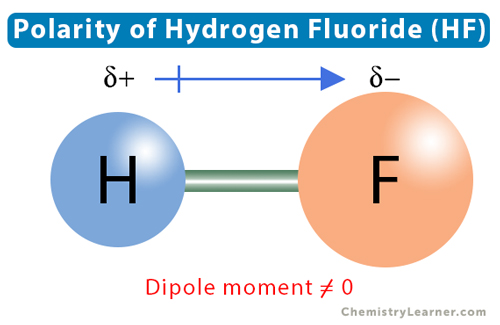
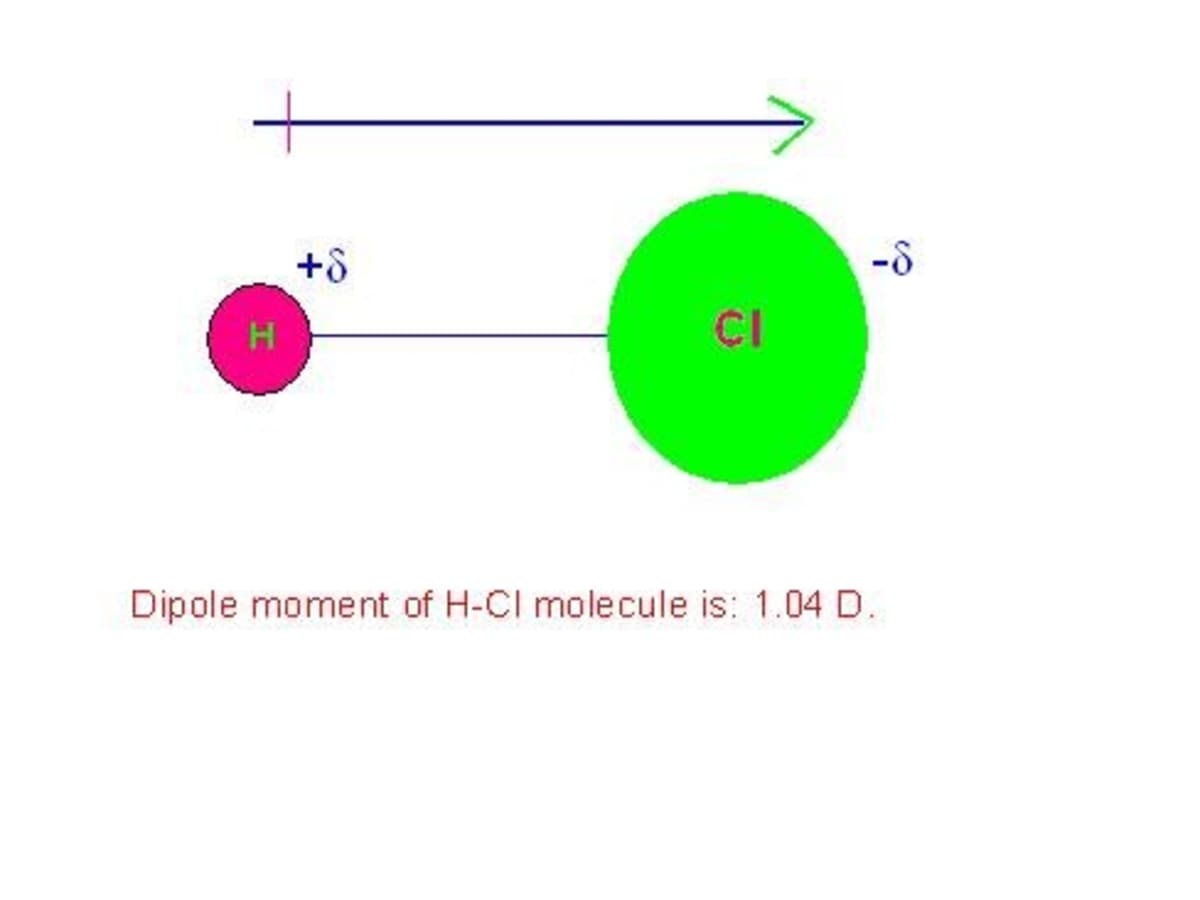
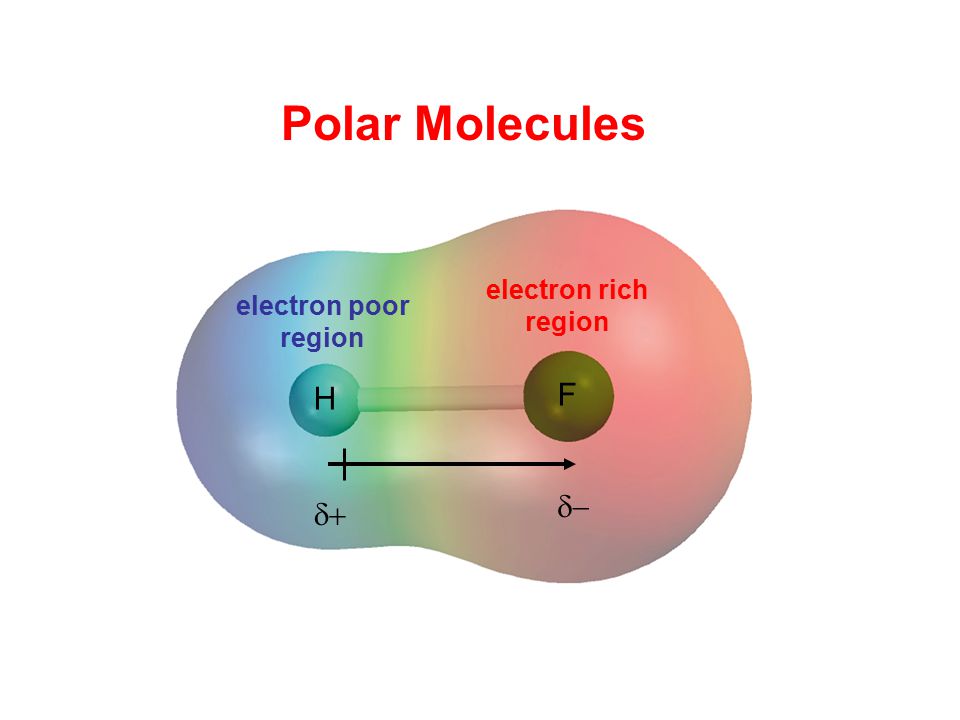
Dipole Moments and Polar Molecules 10.2 H F electron rich region electron poor region = Q x r Q is the charge r is the distance between charges. - ppt download

Write and by the appropriate atoms and draw a dipole moment vector for any of the following molecules that - brainly.com

nar length increases #. The decreasing order of dipole moments of the molecules HF, H20, BeF2, NF3 is a. HF > H2O > BeF, > NFZ bo H2O > HF > NF3 >

Potential energy and transition dipole moment curves of HF. PECs of the... | Download Scientific Diagram
Why is it so that the dipole moment of HI < HBr< HCl<HF, where HI is lowest and HF is highest? - Quora

Values of the dipole moment coefficients of HF in Debye (numbers in... | Download Scientific Diagram
Why is it so that the dipole moment of HI < HBr< HCl<HF, where HI is lowest and HF is highest? - Quora

Calculated electric dipole moments of the HF and LiF diatomic fragments... | Download Scientific Diagram

Qu. The dipole moment of HF je 1.780 bond length of H-F is o2 pm. Calculate the ionic character in Hof. U - 1.78070 in x100

SOLVED:The dipole moment of HF is 1.83 D and the bond length is 91.7 pm . Calculate the amount of charge (in electronic charge units) on the hydrogen and the fluorine atoms

SOLVED:Explain why HCI has a smaller dipole moment than HF, even though the H-Cl bond is longer than the H-F bond.

Values of the dipole moment coefficients of HF in Debye (numbers in... | Download Scientific Diagram