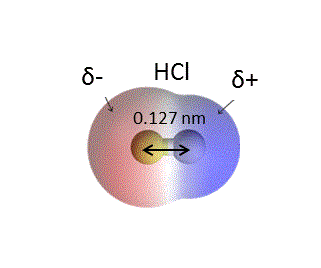
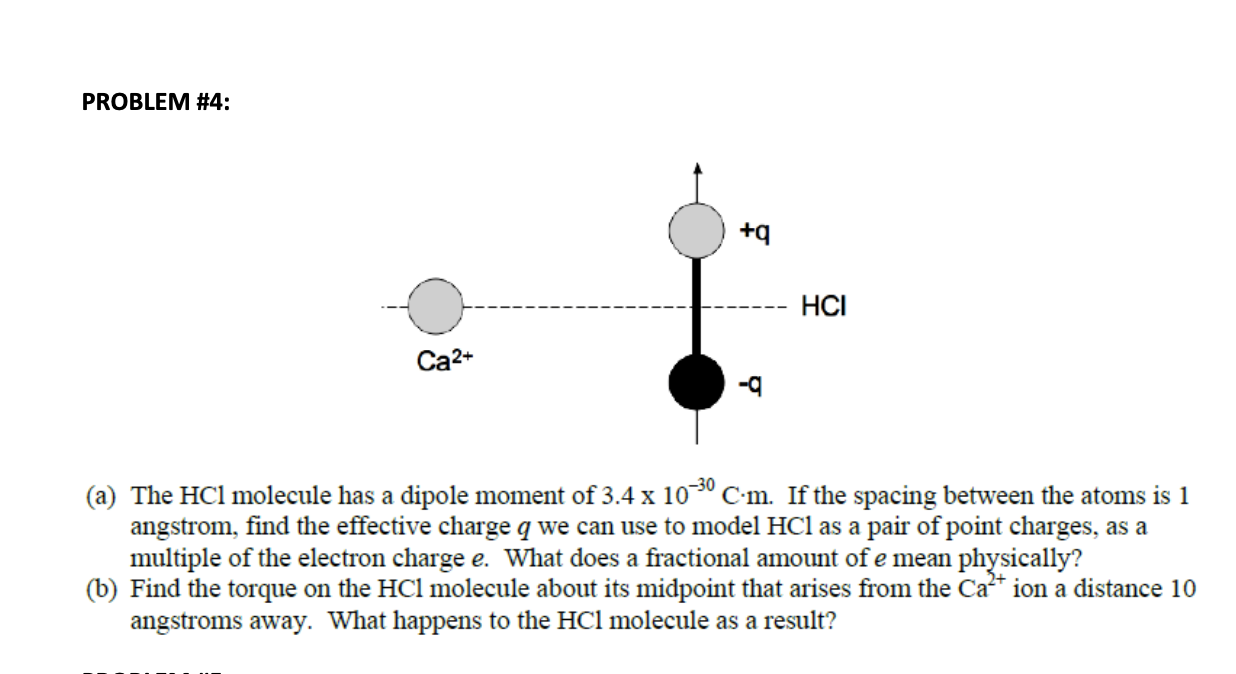
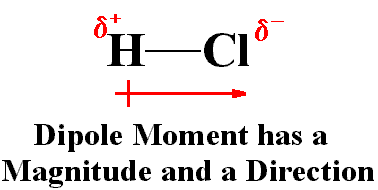An HCl molecule has a dipole moment of `3.4xx 10(-30) Cm` . Assuming that equal and opposite cha... - YouTube

SOLVED: Dipole moment (μ) Polarizability volume " HCl 1.08 D 2.63x10^-30 m^3 HI 0.42 D 5.45x10^-30 m^3 Q1. A) For a given electric field, what is the ratio of the induced dipole
In HCl molecule, expected value of dipole moment is 6.12 D but experimental value is 1.03 D. Then, the percentage ionic character will be Options: a) 16.83 b) 15.14 c) 6.02 d) 18.90
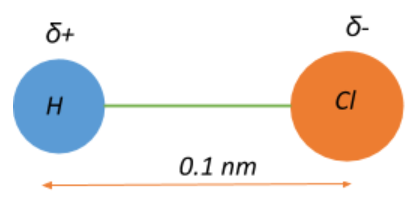
The electric dipole moment of an HCl atom is $3.4 \\times 10^{-30}\\;Cm$. The charges on both atoms are unlike and of the same magnitude. Magnitude of this charge is ______. The distance

20., The observed dipole moment of HCl is 1.03 D.Bond length is 1.275 A then the percentage ofionic - Brainly.in

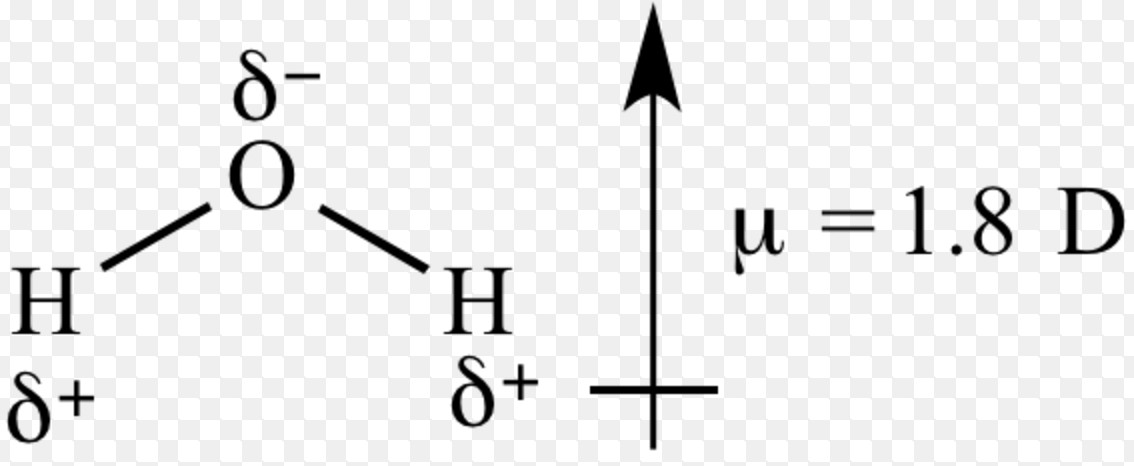

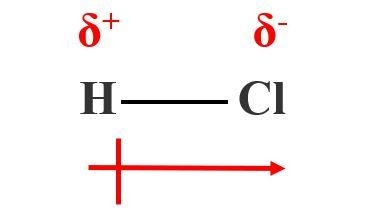
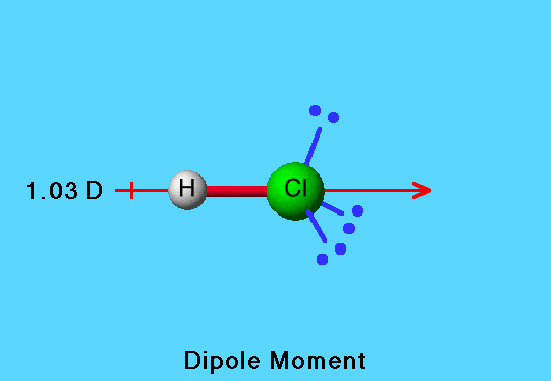

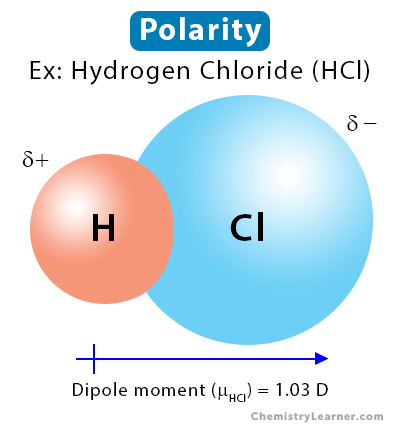

![Best Overview: Is HCl polar or nonpolar [#1] - Science Education and Tutorials Best Overview: Is HCl polar or nonpolar [#1] - Science Education and Tutorials](http://sciedutut.com/wp-content/uploads/2021/06/HCl-cover-banner.png)