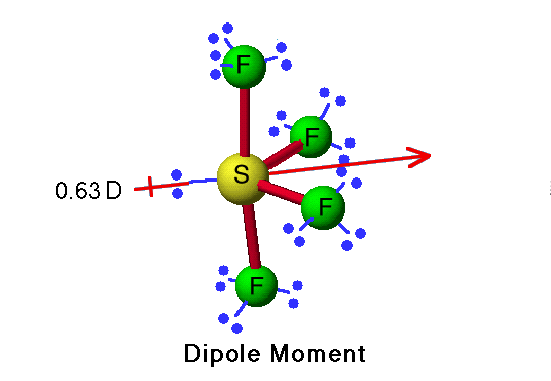
Which have a molecular dipole moment? (Select all that apply.) 1. BF3 2. SF4 3. BrF3 4. NF3 5. CF4 | Homework.Study.com

Who has high dipole moment - SF4 or CF4 pl ans - Chemistry - Coordination Compounds - 13090037 | Meritnation.com
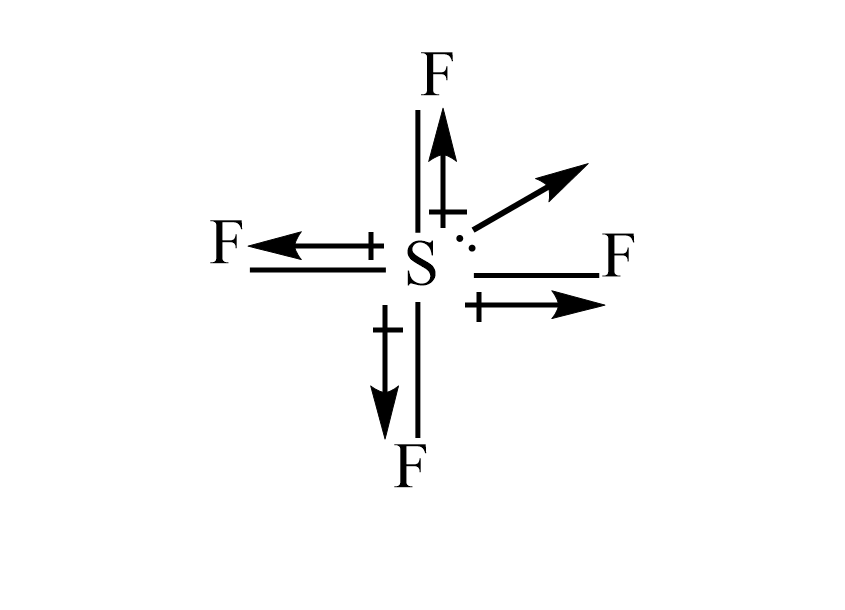
Molecule having a non zero dipole moment?a) $ S{F_4} $ b) $ Si{F_4} $ c) $ Xe{F_4} $ d) $ B{F_3}^{} $
✓ Solved: Explain why CF4 and XeF4 are nonpolar compounds (have no net dipole moments) while SF4 is polar...

Which have a molecular dipole moment? (Select all that apply.) 1. BF3 2. SF4 3. BrF3 4. NF3 5. CF4 | Homework.Study.com
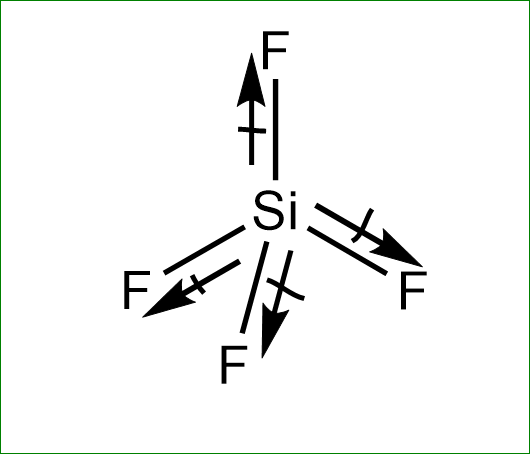
Molecule having a non zero dipole moment?a) $ S{F_4} $ b) $ Si{F_4} $ c) $ Xe{F_4} $ d) $ B{F_3}^{} $
✓ Solved: Explain why CF4 and XeF4 are nonpolar compounds (have no net dipole moments) while SF4 is polar...